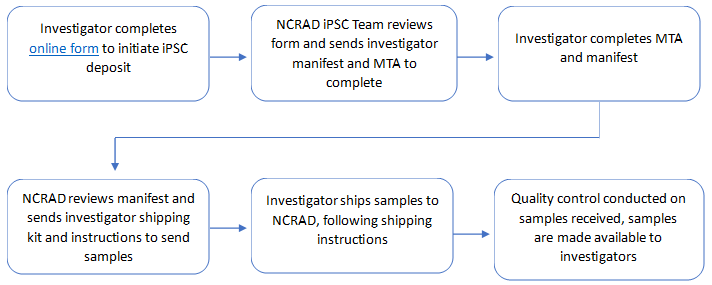
Procedure for Depositing iPSCs and Fibroblast lines to NCRAD
To facilitate sample sharing and increase the rate of research discovery, the National Centralized Repository for Alzheimer's Disease and Related Dementias (NCRAD) is banking induced pluripotent stem cells (iPSCs) and fibroblasts. In order to deposit iPSC and fibroblast specimens, investigators need to first complete and submit the online iPSC & Fibroblast Information Collection form.
This form requires the investigator to provide information and provide documentation that includes:
- Basic information about the investigator.
- Specific information about iPSC lines to be deposited.
- Specific information about fibroblast lines to be deposited.
- Fibroblasts associated with iPSC lines are requested to be deposited with the iPSC lines.
- It is preferred that investigators submit 3 vials per iPSC and fibroblast line. This ensures the lab has extra vials in the event of a QC measure failure. Sending less than 3 could result in additional requests for any vials that fail QC.
- Lines with less than 20 passages are preferred. Lines with greater than 20 passages will be moved to the end of the queue and expanded last.
- Provide documentation of mycoplasma testing and karyotyping for iPSCs and fibroblasts to be deposited, if available.
- Please provide any protocols used for iPSC and fibroblast cell culture.
Once the iPSC & fibroblast information has been submitted, the NCRAD iPSC team will review the information and contact the investigator with any questions, or request additional documentation. Once the NCRAD iPSC team approves, the investigator will be contacted and provided with material transfer agreement (MTA) documents which are not subject to alteration. After completion, the investigator will be supplied with a shipping kit and instructions to send specimens to NCRAD. Once lines are received, the laboratory staff will conduct a host of rigorous quality control measures.
Currently, there is no cost to deposit your lines at NCRAD.
The lines you deposit will be made available to other investigators to increase the rate of research discovery.
The following paperwork is required to be completed before we can accept cell lines:
- iPSC & Fibroblast Information Collection form
- Material Transfer Agreement (MTA)
- A completed manifest
The NCRAD stem cell lab has designed standardized protocols to ensure uniformity and quality control for every line we bank. As such, we require detailed information about your cells to ensure they are transitioned into our laboratory with minimal distribution to the cells and also to be able to provide other investigators with detailed information about the lines.
NCRAD banks iPSCs, gene edited isogenic iPSCs, and associated fibroblasts. Please send all cell types when possible.
No testing is required. However, please provide documentation of mycoplasma testing and karyotyping for iPSCs and fibroblasts to be deposited if available.
It is preferred that investigators submit 3 vials per iPSC and fibroblast line. This ensures the lab has extra vials in the event of a QC measure failure. Sending less than 3 could result in additional requests for any vials that fail QC.
NCRAD will send a shipping kit with instructions prior to shipping, at no cost, that will include everything needed to ship, except pelleted dry ice. You will need to supply pelleted dry ice for shipping. Please do not send samples using your own shipping materials.
Our group of talented iPSC technicians will thaw lines and conduct a host of rigorous quality control measures. Only lines that pass all quality control testing will be expanded and made available for distribution.
The goal of the National Centralized Repository for Alzheimer's Disease and Related Dementias (NCRAD) is to support research focused on the etiology, early detection and therapeutic development for Alzheimer's disease and related dementias. In achieving this goal, it is also top priority to protect research participant privacy. When sending lines, researchers will not be asked/will not provide identifying research participant information with the lines deposited.
Yes, investigators that contribute samples to NCRAD will be able to request samples at a reduced cost.